ZENITH™
La science de la forme parfaite
Precision science. Refined formulation. Designed to support modern metabolic care.
Advancing the Science of Perfect Form
Zenith Bioform is a USA-based specialty company dedicated to developing and supplying advanced formulations that support metabolic health and body composition management.
Guided by the principle “La science de la forme parfaite”, we combine rigorous quality standards with innovative science to provide clinicians with reliable products for modern aesthetic and wellness practice.
Our operations and logistics are based in Reno, Nevada, enabling efficient distribution and responsive support for qualified healthcare professionals.
SCIENCE BEHIND ZENITH™ TIRZEPATIDE
Dual Agonist Mechanism
Zenith Tirzepatide is built on one of the most advanced metabolic science platforms available today: dual incretin activation. Unlike traditional single-pathway peptides, tirzepatide simultaneously targets two key hormonal receptors—GIP and GLP-1—resulting in a synergistic metabolic effect. Its mechanism is associated with:
•Improved insulin sensitivity
• Enhanced postprandial glycemic control
• Delayed gastric emptying
• Appetite regulation and caloric reduction
• Support for adipose tissue metabolism
• Cardiovascular & Anti-Inflammatory Effects
• Decreases in systolic blood pressure
• Potential Anti-Aging & Longevity
(Support Mechanistic information is provided for scientific understanding only and does not constitute prescribing guidance.)
Engineered for Excellence: Zenith’s High-Standard Tirzepatide
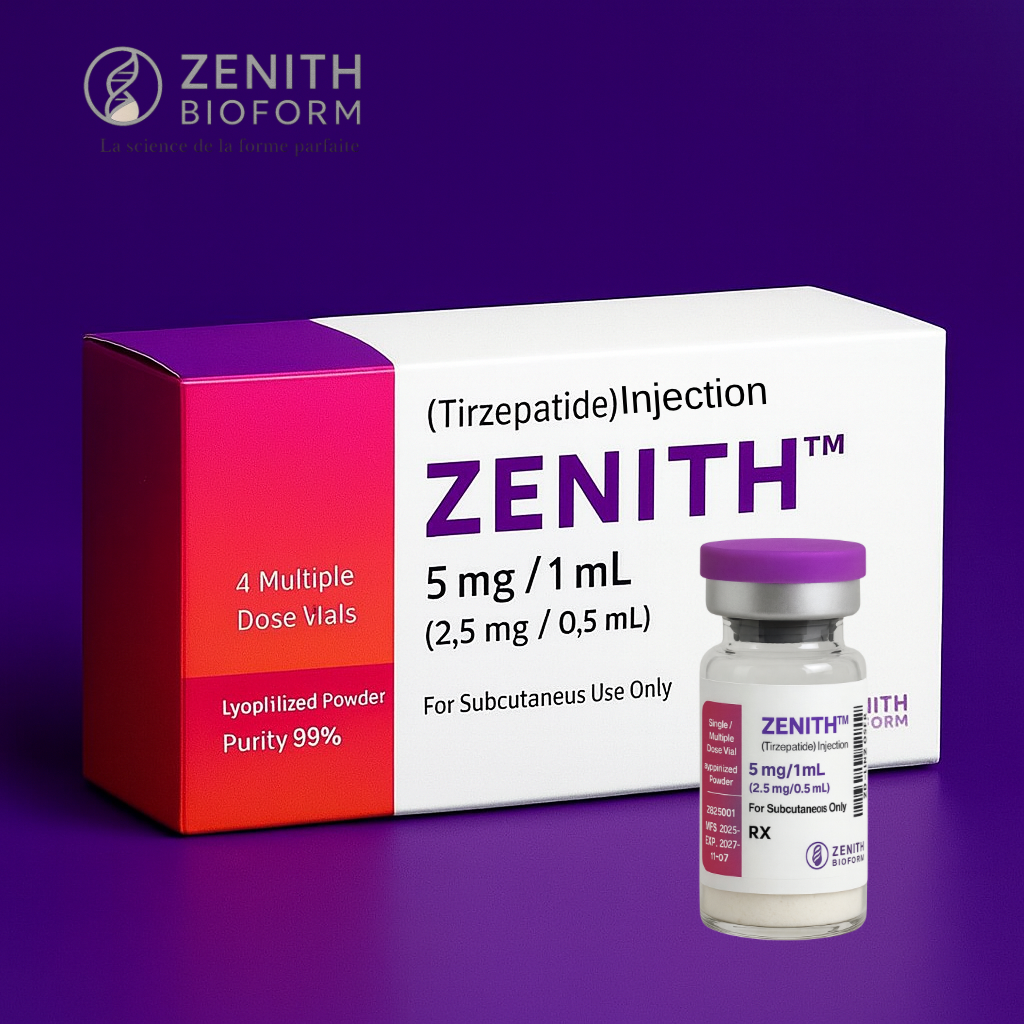
Zenith Tirzepatide is engineered to higher standards than low-quality compounded forms. With 99% purity, GMP-aligned manufacturing, professional lyophilization, precision dosing, and full traceability, Zenith provides clinics with a consistent, reliable, and high-integrity tirzepatide formulation designed for professional use only.
Indication
FDA-Approved Indications
A. Type 2 Diabetes Mellitus (T2DM)
Brand: Mounjaro® Purpose
• Improve blood sugar control
• As monotherapy or combined with other antidiabetic medications
• Helps reduce A1c and aids in weight loss as a secondary effect
Key benefits:
• Strong glucose lowering
• Weight reduction
• Cardiometabolic risk improvement
B. Chronic Weight Management (Obesity / Overweight)
Brand: Zepbound™ Approved for patients with:
• BMI ≥ 30 (obesity)
or
• BMI ≥ 27 with at least one weight-related condition, such as:
• Hypertension
• Type 2 diabetes
• Dyslipidemia
• Obstructive sleep apnea
• Cardiovascular risk factors
Purpose:
Long-term weight management combined with diet and exercise.
Widely utilized in clinical practice and supported by growing evidence.
A. Prediabetes / Insulin Resistance To prevent progression to diabetes.
Zenith Tirzepatide helps manage poor eating patterns through appetite control and improved fullness signals.
Indication, Safety & Warnings, and Adverse Effects For Zenith Tirzepatide
B. Metabolic Syndrome
Helps with:
Weight loss
Insulin resistance
Menstrual irregularity
C. Polycystic Ovary Syndrome (PCOS)
Improves:
Blood pressure
Triglycerides
HDL levels
D. Obstructive Sleep Apnea (OSA) — Weight-Related
Zenith reduces OSA severity through weight loss. (Not a primary treatment—adjunct only.)
Most people with obstructive sleep apnea (OSA) have it worsened by excess body weight, especially fat around the neck and upper airway. Zenith causes significant weight loss, which can improve:
• Airway space
• Breathing during sleep
• Frequency and severity of apneas
• Daytime sleepiness
Some recent studies show that people who lost a large amount of weight with tirzepatide had improvements in OSA severity scores.
But important:
• Zenith Tirzepatide is not a first-line therapy for OSA.
• It does not replace CPAP, mouthguards, or airway treatments.
• Not everyone with sleep apnea is overweight, so tirzepatide won’t help in those cases.
E. Non-Alcoholic Fatty Liver Disease (NAFLD / NASH)
Improves liver fat and inflammation.
F. Cardiovascular Risk Reduction
Due to weight loss, BP lowering, and glycemic improvement.
G. Postpartum Weight Retention For eligible non-breastfeeding mothers.
H. Clinical uptake of tirzepatide is widespread in the USA, Europe, the Middle East, and Southeast Asia, underscoring its rising role in modern metabolic care.
Body reshaping / medical weight loss
Visceral fat reduction
Appetite control therapy
Improved metabolic aging
Safety and Warnings
To protect patients and clients safety, the following precautions must be observed:
General Use Warnings
For professional use only
Not for self-administration
Not recommended for individuals with severe gastrointestinal disease, chronic pancreatitis, or known endocrine disorders unless medically evaluated
Not for use during pregnancy or breastfeeding • Not to be combined with other GLP-1 agonists unless directed by a licensed clinician
Discontinue immediately if severe gastrointestinal symptoms, dehydration, or signs of pancreatitis occur
Contraindication Warnings
Zenith should not be used in:
patients with a personal or family history of medullary thyroid carcinoma (MTC)
patients with Multiple Endocrine Neoplasia syndrome type 2 (MEN 2)
individuals with known hypersensitivity to peptide components/allergy to tirzepatide
individuals with uncontrolled endocrine or metabolic disorders without clinician clearance
Professional Monitoring Recommended Clinicians should monitor:
gastrointestinal tolerance
hydration status
potential gallbladder symptoms
renal function in at-risk patients
overall metabolic response
dose-related tolerability during titration
Adverse Effects
Based on published clinical research on tirzepatide, the following adverse reactions have been commonly observed:
Common (Usually Mild to Moderate)
Nausea
Vomiting
Diarrhea
Constipation
Decreased appetite
Bloating or indigestion
Fatigue
Dizziness
Injection-site redness or irritation Less Common
Dehydration from persistent GI symptoms
Abdominal discomfort
Reflux or delayed gastric emptying sensations Rare but Serious (Medical Attention Required)
Severe abdominal pain (possible pancreatitis)
Signs of gallbladder disease
Persistent vomiting leading to dehydration
Allergic or hypersensitivity reactions
Severe dizziness or weakness
Any persistent, worsening, or severe symptoms should be addressed immediately by a licensed healthcare professional.
Protect Yourself from Counterfeit and Unsafe, Mass-Compounded Products
Counterfeit or poorly compounded tirzepatide products can pose serious risks due to inconsistent potency, contamination, and lack of proper testing. To safeguard your health, always choose formulations prepared under licensed 503A compounding pharmacies. Zenith Bioform partners exclusively with facilities that follow strict FDA and state pharmacy regulations, ensuring full traceability, batch verification, and 99%+ purity testing for every vial. This commitment to regulated standards helps protect clinics and patients from unsafe, unverified, or mass-produced alternatives.
☑ Before You Begin: Safety Checklist
Review the following with your licensed healthcare provider before starting Zenith Tirzepatide:
Health Conditions
☐ Any pancreas-related conditions
☐ History of severe stomach issues or delayed gastric emptying (gastroparesis)
☐ Digestive problems or difficulty tolerating certain foods
☐ History of diabetic eye complications (retinopathy)
Medications You Currently Take
☐ Insulin or sulfonylureas
☐ Any other prescription medications
☐ Over-the-counter drugs or supplements
☐ Herbal or natural products Lifestyle & Upcoming Procedures
☐ Scheduled surgery or procedures requiring anesthesia or sedation
☐ Major changes in diet or wellness routines planned Pregnancy & Breastfeeding
☐ Currently pregnant or planning to become pregnant
☐ Currently breastfeeding or planning to breastfeed
☑ Checklist for Safe Use
☐ Use Zenith only under professional supervision
☐ Follow the dose and schedule your provider recommends
☐ Injections are subcutaneous only (abdomen, thigh, upper arm)
☐ Do not mix Zenith with insulin or any other injectable medication
☐ Change/rotate injection sites weekly
☐ Keep doses on the same day each week
☐ Contact a provider immediately if too much is administered or if unexpected symptoms occur
Zenith Tirzepatide — Dosing & Titration Guide
Lyophilized Powder • ≥99% Purity • 4 Vials per Box
Available vial strengths: 5 mg • 10 mg • 15 mg
Zenith Tirzepatide is a high-purity, sterile lyophilized formulation designed for professional use in clinical and wellness programs. All dosing, titration, and administration must be supervised by a licensed healthcare provider.
📈Standard Weekly Titration Schedule
Titration helps improve comfort and minimize gastrointestinal side effects. Providers may adjust based on individual tolerance.
Weeks 1–4
2.5 mg once weekly Introductory dose for tolerability.
Weeks 5–8
5 mg once weekly ,First active dose. (Using 5 mg vial)
Weeks 9–12
7.5 mg once weekly , intermediatetitration stage. (Prepared from 10 mg vial as directed)
Weeks 13–16
10 mg once weekly Enhanced metabolic support. (Using 10 mg vial)
Weeks 17–20
12.5 mg once weekly Advanced titration. (Prepared from 15 mg vial)
Week 21 and onward
15 mg once weekly Maximum standard dose. (Using 15 mg vial)
Storage Instructions (Unreconstituted Vials)
• Store lyophilized vials refrigerated at 2–8°C (36–46°F).
• Keep vials protected from light and in original packaging until use.
• Do not freeze the product.
• Avoid exposure to heat, direct sunlight, or temperature fluctuations.
• Always check the lot number, appearance, and expiry before use.
Storage After Reconstitution
Once reconstituted with sterile water for injection (SWFI):
• Use immediately or store refrigerated at 2–8°C. • Do not freeze reconstituted solution.
• Use within 28 days if kept refrigerated.
• Discard the vial if:
• the solution becomes cloudy, discolored, or contains particles
• sterility is compromised
• The vial has been stored outside temperature guidelines
Vial Strength Typical Use
5 mg vial Starter and early therapeutic dosing
10 mg vial Intermediate dosing / escalation
15 mg vial Advanced or maintenance dosing
📌 Administration & Handling Notes
• Inject once weekly, on the same day each week (as directed by the provider).
• Zenith Tirzepatide is administered subcutaneously only.
• Rotate injection sites to avoid irritation.
• Do not mix tirzepatide with insulin or any other injectable product.
• Reconstitute only with sterile water for injection (SWFI).
• Gently swirl—do not shake—until clear. • Use aseptic technique throughout preparation and handling.
Monthly Patient Program Pricing
Common USA ranges for wellness/medical clinics:
• Entry Dose Program (2.5 → 5 mg): $350 – $550 / month
• Mid-Range Program (7.5 → 10 mg): $450 – $750 / month
• High-Dose Program (12.5 → 15 mg): $650 – $950 / month
📌 When to Adjust or Pause Titration
Your provider may adjust your dose schedule if you experience:
• persistent nausea
• abdominal discomfort
• vomiting • bloating or early fullness
• dizziness or dehydration. Titration should be personalized based on tolerability.
📌 Professional Use Notice
Zenith Tirzepatide is intended exclusively for licensed medical professionals and clinically supervised programs. Not for unsupervised or over-the-counter use.
Zenith™ The science of the perfect form.
Follow us for updates on research, clinical applications, and product advancements: • Email Newsletter